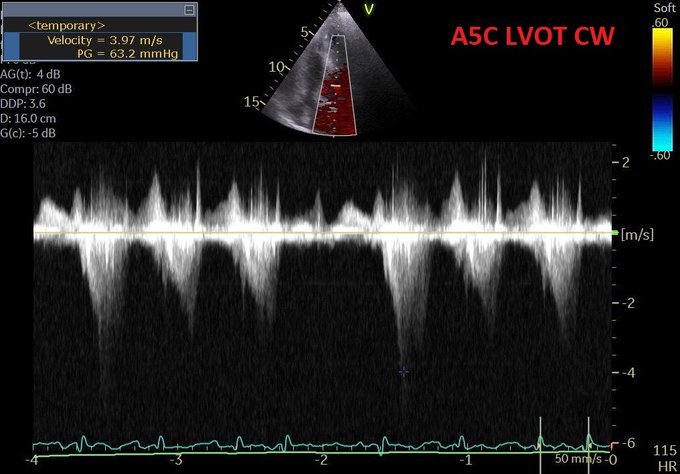
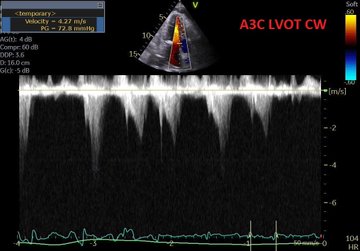
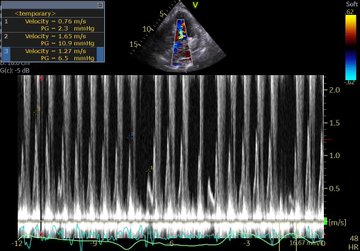
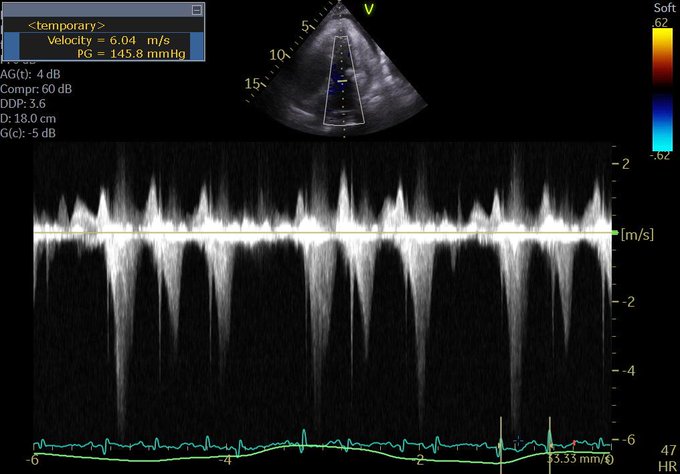
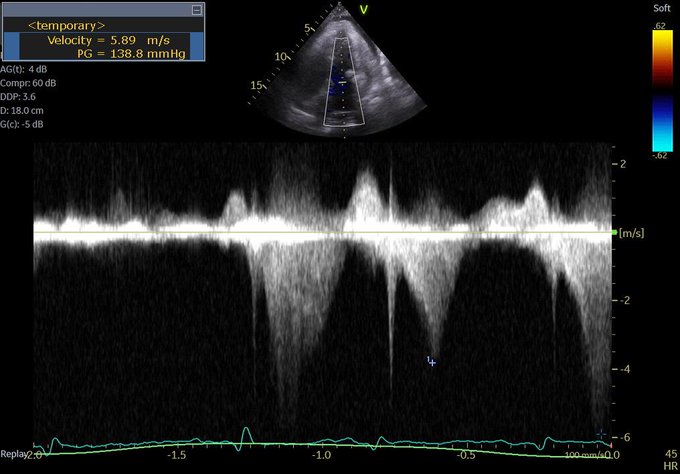
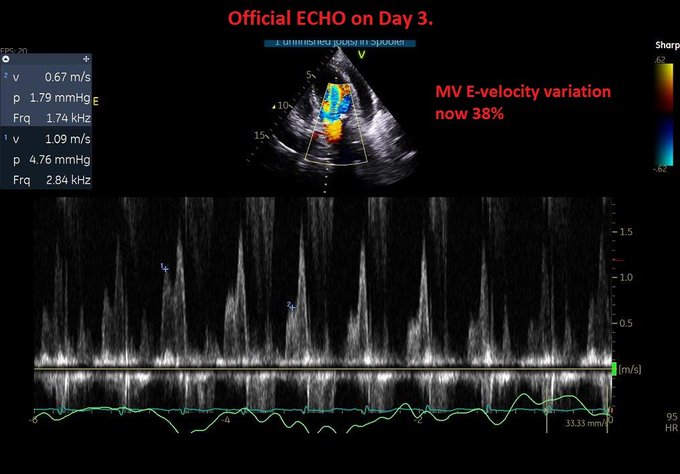
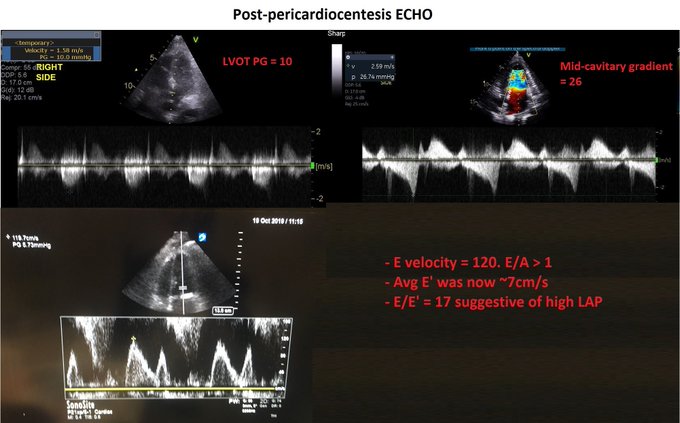
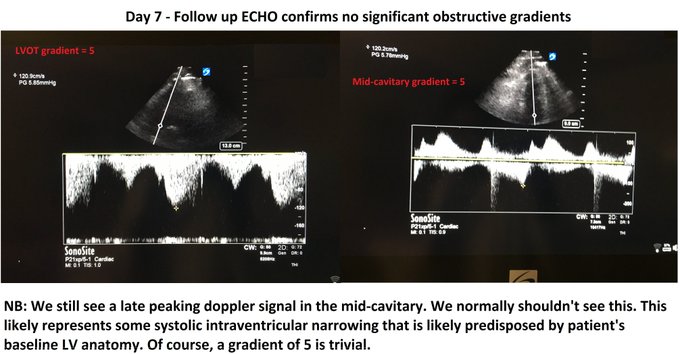
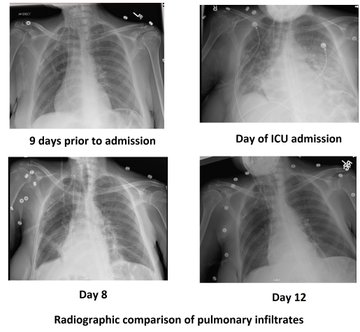
Perhaps this slowed up (0.5x) A3C loop will help with that LVOT doppler!
tamponade
Emergency Pericardiocentesis post-arrest (Part 1). #FOAMed, #FOAMus, #FOAMer
So a few nights ago I got pulled out of slumber to rush to the ER for an elderly patient who had arrested in hospital shortly after having been brought in for chest pain. The sharp ER doc had diagnosed a tamponade on a presumed aortic dissection, managed to get a needle in, aspirated some fluid and managed to get ROSC.
So when I got there we had a patient post-ROSC in rapid atrial fibrillation with a thready but palpable pulse. POCUS showed a large pericardial effusion with minimal LV filling. So here is what we did:
With the catheter in, we were able to drain. Note a couple of POCUS teaching points, always make sure to (1) visualize your guidewire in the right space, and (2) second, when using a dilator, you can note the disappearance of the proximal part of the guidewire as it is covered by the dilator. This tells you you have adequately dilated into the target structure – pericardium in this case, because it is possible (personal experience) to advance a dilator fairly deep, but not go through a perhaps fibrotic pericardium, and then result in pigtail mis-placement just outside of the target.
In part 2 you can also see the aspiration of the effusion and improved LV filling. The patient’s BP instantly rose to 140’s systolic.
More case details and POCUS teaching points to come in part 2.
cheers,
ps – a sterile probe cover was unavailable immediately in the ER. By the time it showed up the pigtail was in. We didn’t feel we could wait. We doused it in alcohol.
Philippe
Bedside US Procedure: Pericardial drainage – Pearls! #FOAMed, #FOAMcc, #FOAMus
So here is a video of a pericardial drain placement for pre-tamponade in a 33 yr old man, presumptively for a viral pericarditis (cultures and cytology pending). In this case, the approach was subxiphoid, because this offered a large pocket of fluid with little or no risk of hitting the RV. The apical approach would have been more risky. Due to technical issues, the video only starts once the guide wire is already in place, but there are a couple of teaching points worth sharing nonetheless.
First, it is useful to confirm guide wire placement prior to dilating. Secondly, in cases such as this where the distance to the pericardium is more than a couple of centimetres (it was about 6 cm here), it is nice to be able to confirm under real-time that the dilator is indeed in the intended area. Because the guide wire is highly echogenic, and the dilator is not, one can see the proximal part of the guide wire “disappear” which indicates that the dilator has covered it, now visibly in the effusion. Once the pigtail is inserted over the guide wire, final confirmation can be obtained by injecting back thru the pigtail and seeing echogenic material (due to minute amounts of air) appear in the pericardium. This is known as the Ajmo sign.
Cheers!
Philippe
Pericardiocentesis for tamponade w/bedside ultrasound: Procedure Video. #FOAMed, #FOAMcc, #FOAMus
So this case was interesting on a couple of levels.
A 76 year old woman presented to the ER with a complaint of abdominal discomfort and was admitted with a diagnosis of pneumonia and lower abdominal cellulitis. She had a history of diabetes, obesity and remote oral cancer which had been treated 6 yrs ago. The next morning, while still in the ER awaiting a ward bed, she had a hypotensive episode, and fortunately the ER doc on shift grabbed an ultrasound probe and took a look, calling me a few minutes later with a diagnosis of tamponade. She was absolutely correct. I saw and echo’d her shortly after:
The first two clips show the IVC, which is distended with minimal variation. This should prompt the bedside sonographer to anticipate downstream pathology (except for iatrogenic volume overload and renal failure…).
The subsequent clips show subxiphoid views (and one clip of the associated left pleural effusion) showing a significant pericardial effusion and difficult to distinguish cardiac chambers.
Clinically, she was dyspneic, uncomfortable, HR 115, BP 130’s systolic (in ER in 80’s then got some fluid). Her heart sounds were not particularly quiet, and her JVP was difficult to assess due to obesity.
Here is the drainage video:
Her abdominal pain resolved very rapidly, her breathing improved and vitals stabilized.
Pathology is still pending, but bloody effusions commonly include malignancy, tuberculosis, but also simple viral paricarditis.
So I think this is a great case for the argument of integrating ultrasound into physical examination rather than as an ancillary test. Because she didn’t present with a predominant hypotensive or respiratory component, the diagnosis wasn’t seriously entertaine, and obesity, body habitus and pleural effusion undoubtedly made physicians overlook the cardiomegaly. However, in my opinion and that of most bedside sonographers, abdominal pain warrants an abdominal us exam, and the distended IVC would have prompted at least a quick cardiac assessment, and the effusion would have been noted immediately.
In my CC/IM practice, hardly anyone escapes the probe, as cardiopulmonary and abdominal status is hardly ever irrelevant to me…
cheers!
Philippe
Bedside Ultrasound in Cardiac Arrest: A Quick Overview, ISURA 2014. #FOAMed, #FOAMcc, #FOAMus
Here’s a quick lecture I gave at Vincent Chan’s ISURA 2014 in Toronto. It was a great event attracting anasthetists from all over the world, and it was an honor to work with Massimiliano Meineiri, Alberto Goffi, Adriaan Van Rensburg, Colin Royse and many more.
Unfortunately it was my first time using Prezi, which is pretty cool but doesnèt allow you to loop your videos, which really, really sucks! So unfortunately the clips are very short…
Love to hear any comments!
Philippe
Bedside ultrasound clip quiz 1 – #FOAMed, #FOAMcc
62 year old with weight loss, tachycardia and progressive dyspnea…what do you see?
scroll below!
This was a case of pre-tamponade/tamponade. Here is a view a few minutes later, with the guidewire in. This was a case of malignant pericardial effusion.